My Cart
Your Shopping Cart is currently empty. Use Quick Order or Search to quickly add items to your order!
 Hedi Baxter Lauffer, PhD
Hedi Baxter Lauffer, PhD
Director of the Wisconsin Fast Plants® Program
University of Wisconsin-Madison
February 2016
Middle and high school students need an opportunity to construct evidence-based explanations for how variation and natural selection can lead to adaptation of populations over time (NGSS MS-LS4-4 and HS-LS4-4). However, managing a population of classroom-friendly living organisms that consistently grow, develop, and thrive while students observe variation among individuals can be a real challenge.
Another challenge can be finding a model organism that responds quickly to environmental variables, allowing students to gather relevant evidence that some individuals are better able to survive when conditions change. Wisconsin Fast Plants® offer solutions to these challenges with several fast-growing options that are easy to manage and involve an organism large enough to observe with the unaided eye or a simple hand lens.
Let me share with you a technique for engaging students with a population of 5- or 6-day-old Fast Plants® seedlings. Fast Plants® can now be planted and reared using ready-made seed disks in a deli container growing system. This makes using them for variation and natural selection lessons easier than ever. With this technique, students can observe variation in their seedling population, subject it to an abiotic environmental change—salinity in this example—and see the effects. Using evidence collected from their population, students explain their hypotheses for how natural selection could change a Fast Plants® population over time if it was subjected to environmental conditions like the salt contamination their seedlings experienced.
Salt (mainly sodium chloride, NaCl) is introduced into the environment in many different ways and can cause a wide variety of problems. In the northern US, salt is frequently used to melt ice on roads. Most of this salt ends up in roadside soils. Overhead irrigation can cause the buildup of salts on farm fields. This is due to the irrigation water evaporating and depositing its dissolved salts on the fields. These are examples of salt contamination.
In coastal areas, extensive pumping from wells in groundwater aquifers can cause the infiltration of salts from the oceans into the aquifers and related soils. This is an example of salt infiltration. Through the use of a deli container growing system and Fast Plants®, it is possible to model some of the effects of salt contamination and infiltration on the environment. Fast Plants® are a good choice for this because they respond quickly to the presence of salt.
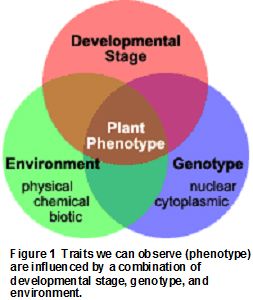 Variation of a particular visible trait (phenotype) among individuals in a population is caused by a combination of the genetic makeup (genotype) of the individuals and their environment. Three broad categories of environmental components interact with an individual’s genotype to influence what we observe as their phenotype: the physical environment, the chemical environment, and the biological environment. See Fig. 1. Fast Plants® are particularly well suited for variation investigations because they reproduce sexually and do not self-pollinate. Therefore, every individual in a population of Fast Plants® has a unique genotype—much like human beings.
Variation of a particular visible trait (phenotype) among individuals in a population is caused by a combination of the genetic makeup (genotype) of the individuals and their environment. Three broad categories of environmental components interact with an individual’s genotype to influence what we observe as their phenotype: the physical environment, the chemical environment, and the biological environment. See Fig. 1. Fast Plants® are particularly well suited for variation investigations because they reproduce sexually and do not self-pollinate. Therefore, every individual in a population of Fast Plants® has a unique genotype—much like human beings.
To identify variation, a trait must be observed, described, and measured or quantified. For example, to investigate variation in the trait plant height in a population of Fast Plants®, the height of all individual plants in the population (at the same age) is recorded and organized (graphed). The variation of the trait in the population is then expressed as the variety of heights that exist within a range, from the shortest to the tallest.
Another way to view variation is to determine whether the trait is present or absent. For example, the purple color in Fast Plants® stems may be present or absent and, if it is present, the intensity of the purple may vary. Other traits vary in their degree of expression or numbers. Students can, for example, observe in a Fast Plants® population that the total number of hairs on the stems and leaves varies considerably from plant to plant. Just as with height, the number of hairs can be quantified by counting and graphing them to show the extent of variation in the Fast Plants® population’s hairiness.
Variation occurs at all stages of the life cycle. In this investigation, students observe 3- or 4-day-old Fast Plants® seedlings to identify traits expressed with variation. For example, hypocotyl length (distance between roots and the cotyledons or seed leaves), cotyledon width, root length, the presence or absence of purple stem color (and its intensity) are all easily observable traits that vary.
| 1. | Introduce the leading questions: What variations in traits can you observe in your population of Wisconsin Fast Plants® seedlings? What causes those variations and what advantages can they have for individuals or for the population? Explain how students will work in small groups to observe and gather evidence about the variation of traits in a Fast Plants® population and relate those observations to the investigation questions. | ||||||
| 2. | Students working in small groups plant 2 seed disks in 2 deli container growing systems, add water, and place them under bright light to germinate. In 3 to 4 days, seeds will have germinated, and seedlings will emerge, showing variation in a variety of observable traits.
|
||||||
| 3. | Five days after planting, pose a follow-up investigation question: How might variation among individuals in your Fast Plants® seedling population give some individuals an advantage in surviving a change in their environment?
|
||||||
| 4. | Salt contamination can be modeled in a deli container growing system by applying a salt solution to the soil surface twice—6 and 9 days after planting. Use a 1% (weight/volume) salt solution as a starting concentration.
|
||||||
| 5. | Salt infiltration can also be modeled in a deli container growing system by adding salt to the aquatic system (water reservoir) that is connected to the terrestrial system above by a capillary wick. A good place to start when investigating infiltration effects is to use the same 1% salt solution that was applied in the contamination experiment as the starting aquatic solution in this system. It may take time for the salt to move into the soil above, so have some patience and keep watch out for initially subtle changes in the growth of plants in the terrestrial system. | ||||||
| 6. | In any experiment, it is important to have a control. In this experiment, each student group grows and observes a control: a parallel deli container growing system that is treated with pure water every time the salt solution is added to the experimental deli container growing system. If different student groups are running a variety of salt treatment methods—such as applying salt from above (contamination) versus below (infiltration)—add equal volumes of salt solution or pure water to each deli container growing system for each treatment. | ||||||
| 7. | Keep close observations on as many Fast Plants® traits and other aspects of the terrestrial and aquatic systems as possible. All sorts of surprises can arise. One that students may observe is the differential loss of water from the reservoir of the systems receiving salt treatments. This is most likely caused by salt-related physiological stress and the resulting reduced transpiration rates. | ||||||
| 8. | Connect students’ observations about survival rates to the overarching investigation question about how some individuals in a population can have traits that are advantageous in certain environmental conditions. Guide students to relate these observations to an evidence-based claim about how the frequency of traits that offer an advantage for surviving salt contamination could change in the population over generations. | ||||||
| 9. | For this experiment, it is valuable to have students grow their populations of Fast Plants® through to seed production. Guide students to continue looking for variation in traits among individuals in their treatment and control populations. Frequently, those plants receiving salt contamination produce less pollen and fewer seeds—some may fail to produce seeds at all. Students who observe this phenomenon have an opportunity to develop a deep understanding of the difference between survival and reproductive fitness. Individual plants that survive salt contamination but cannot reproduce cannot influence the genetic makeup of future generations. |
Browse Carolina.com and www.fastplants.org for more information about teaching and learning with Fast Plants®.
