My Cart
Your Shopping Cart is currently empty. Use Quick Order or Search to quickly add items to your order!
Magnetism is a physical property of certain metals. A particular alignment of their atoms may produce a magnetic field. Within a magnetic field, lines of force can be detected and measured. These lines seem to flow through and around a magnet, out from the north pole, then back through the south pole.
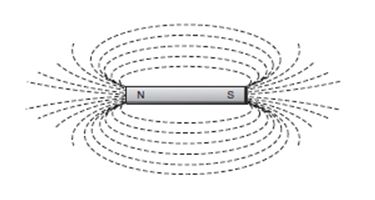
The word magnet comes from the Greek word meaning "the stone of Magnesia." In a region once known as Magnesia, the Greeks found a magnetic iron ore (lodestone) that attracted iron-containing materials. These naturally occurring rocks can be found in various parts of the world. In addition to occurring naturally, magnetism can be induced in ferromagnetic metals (iron, nickel, and cobalt).
Magnets are not just for attaching things to refrigerators. They are all around us—in computer hard drives, speakers, medical equipment, toys, and motors. One peculiar use of magnets involves cows. A special long, cylindrical magnet is administered like a pill to a cow. The fairly heavy magnet remains in the bottom of the cow’s stomach, where it attracts and holds bits of steel and iron that the cow accidentally ingests while grazing (e.g., barbed wire, fence nails). This lessens the likelihood that sharp metal will churn through the stomach until it pokes through and damages another organ (often the heart), causing a condition called hardware disease. Note: Humans should never eat magnets! They can stick together through intestinal walls, causing damage and requiring surgery to remove.
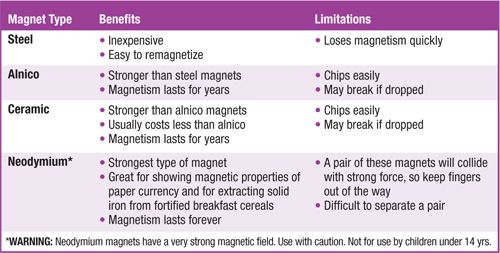
Magnets are made from different materials. Alnico magnets are so-named because they are made from aluminum, nickel, and cobalt. Ceramic magnets contain iron oxide particles embedded in a ceramic matrix. Neodymium magnets are among the rare-earth magnets. See Fig. 2 for some of the comparative features of various types:
Grades 5–8
Science as Inquiry
Physical Science
Science and Technology
Grades 9–12
Science as Inquiry
Physical Science
Materials
Preparation and procedure
Here are 2 additional magnet tests that students enjoy:
Currency
US bills are printed with magnetic ink. The Federal Reserve Banks remove worn paper currency from circulation, shred the bills, and sell the shredded currency. Shredded currency may contain all denominations of bills. In the above procedure, substitute 1 cup of shredded currency for breakfast cereal. Compare the amount of iron found in each. Note: You may want to cut the currency into short pieces. Long pieces may entangle the blender blade.
Fingernail polish
Many stores carry fingernail polish that contains iron particles. Paint a fingernail (or other surface) with polish and, before it dries, quickly position the magnet (found on the product’s cap) close to but not touching the wet polish for 10 to 15 seconds. A pattern will appear. Explore how different types of magnets affect the polish. Challenge the class to explain why the manufacturer’s magnet makes a pattern. Note: Be sure to wrap the magnets with plastic to protect them from the polish. Also, be aware that a neodymium magnet is strong enough to remove iron particles from the polish.