My Cart
Your Shopping Cart is currently empty. Use Quick Order or Search to quickly add items to your order!
By Bruce Wilson
Product Manager, Chemistry
Updated November 2017
Acid-base indicators are weak organic acids. Unlike most acids, however, the acid and base forms of indicators are different colors. Since the color of the indicator depends on the pH of the solution, indicators find wide use in applications that involve pH changes, such as titrations, pH testing, and science demonstrations.
The most important property of an indicator is its pH range, which is dependent on the indicator's acid strength. An indicator's pH range is the range of pH values over which the indicator changes colors from its acid form to its base form. It extends from the highest pH at which only the acid form is visible to the lowest pH at which only the base form is visible. The indicator is not sensitive to pH changes outside its range because the indicator does not change color over these pH values. For example, methyl red does not change color over the pH values 1 to 4 or 6 and above. The pH range is 4 to 6 (see Table 1).
Table 1. Carolina's Guide to Indicators Chart
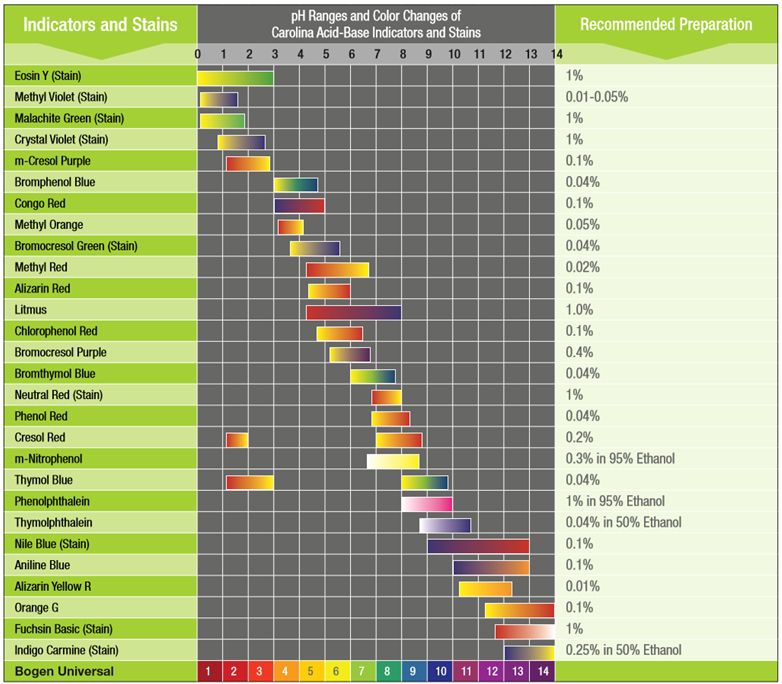
Because a noticeable pH change occurs near the equivalence point of acid-base titrations, an indicator can be used to signal the end of a titration. When selecting an indicator for acid-base titrations, choose an indicator whose pH range falls within the pH change of the reaction. For example, in the titration of a strong acid with a strong base, the pH quickly changes from 3 to 11. The indicator phenolphthalein, whose range spans from pH 8 to 10, therefore makes a good choice for this type of titration.
If you don't know the pH change around the equivalence point of your titration, consult a general chemistry textbook. Most textbooks contain the titration curves for simple titrations, such as weak acid-strong base or strong acid-weak base.
Litmus, extracted from lichen, is one of the oldest and simplest pH indicators. Students may already be familiar with it.
There are 3 types of litmus paper: neutral, red, and blue. Neutral litmus paper is purple; it turns red in the presence of an acid and blue in the presence of a base. Red litmus paper turns blue in the presence of a base. Blue litmus paper turns red in the presence of an acid. Litmus paper does not indicate the pH of a solution, only whether it's an acid or base.
To determine pH, use pH paper, universal indicator solution, or a pH meter. pH meters can detect pH down to 0.01 of a pH unit. This type of precision is usually reserved for advanced science, college, or research coursework. Most general chemistry labs use pH paper or universal indicator solution instead of meters.
pH paper contains several indicators and combines their pH ranges to offer an extended range of pH sensitivity. These papers display a unique color for each pH unit and come with their own color chart.
Universal indicator solutions work similarly and can be the most cost-effective option because only 1 drop is usually required. Bogen universal indicator solution, which contains the indicators methyl red, phenolphthalein, and bromthymol blue, measures pH over the range 4 to 10. Another popular universal indicator solution is also natural, red cabbage juice.
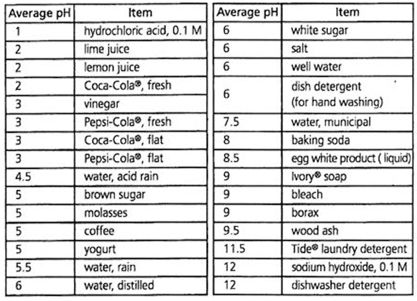
For a fun activity, measure the pH of common household liquids with a pH indicator of your choice. The results may surprise you (see Table 2)!
Table 2. pH of Common Household Acids and Bases
Imagine your students’ surprise when words suddenly appear on a seemingly blank piece of paper sprayed with “water”! What your students don’t know is that the paper was pre-treated with an indicator solution and activated by the pH of the “water” (an acidic or basic solution). The rapid color change of indicators is the “magic” behind many classic “disappearing” ink chemistry demos. To see indicators in action, try this fun activity: